Abstract

Background: AHCT is a standard of care treatment for eligible patients (pts) with MM. Most consensus guidelines recommend that at the time of first AHCT, enough APBHC should be collected to perform two AHCT. However, recent studies have shown that with each decade after 1990, the use of cryopreserved APBHC for a salvage AHCT has declined and is now in the range of 4.6-15% in the decade between 2010-2018. The collection and storage of APBHC for two AHCT incurs additional costs and a prior single center cost analysis revealed that having had collected for just one AHCT in a cohort of 726 MM pts would have saved 3.8 million USD between 1993 and 2011. 1 Most centers that perform AHCT for MM pts continue to collect and store PBAHC for 2 AHCT. Herein we evaluate utilization trends and costs associated with cryopreserved APBHC in patients with MM.
Methods: We retrospectively evaluated the clinicopathologic data from 440 patients with MM who underwent APBHC mobilization and collection at Mayo Clinic Florida between 2010 and 2019. The number of apheresis/collection sessions and APBHC product collection yields were determined for patients who were mobilized by any method. We estimated the costs involved in PBSC collection (apheresis) and cryopreservation storage based on our institution-specific charges as of May 2021. The cost of 1 session of APBHC collection/apheresis was $4,680 and the cost of 1 year of APBHC cryopreservation was $4,790 per patient. Statistical analysis was performed using JMP Pro 15 (SAS). Chi-square and Fischer exact tests were used to carry out univariate analysis for categorical variables and Wilcoxon rank sum/Kruskal Wallis for continuous variables.
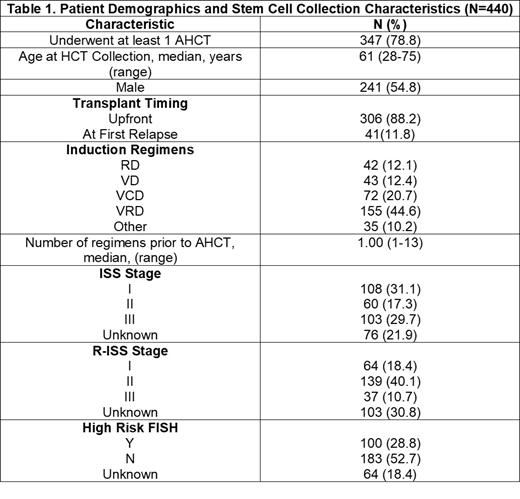
Results: Patient demographics are shown in Table 1. 440 pts underwent at least one collection session and 347 underwent at least 1 AHCT. The median number of mobilization sessions was 1 (95% CI 1.0-1.03), the median number of collection/apheresis sessions was 2.0 (95% CI 2.39-2.59), 84.3% (n=371) required ≥1 collection session. The median number of CD34 stem cells collected was 7.6 x10 6 (95% CI 7.76-8.19 x10 6), and the median number of infused CD34 cells was 4 x10 6(95% CI 4.13-4.37x10 6). The median cost of total collection sessions (defined as # of apheresis/collection sessions x $4,680) was $9,360 per pt (95% CI $11,181-$12,100). The median cost of cryopreservation (defined as # of years of storage x $4,790) was $19,160 per pt (95% CI $9,682-$22,252). The median number of years of storage was 4 (95% CI 4.11-4.65). 77(17.5%) pts have had APBHC in storage for <2 years, 218 (49.5%) have had APBHC in storage for 2-5 years, and 145 (33%) had have APBHC in storage for >5 years. The median time from MM diagnosis to AHCT was 9.0 months (range 13.8-18.5). 82% of patients collected enough APBHC (≥6x10 6, at least 3x10 6 per AHCT)for 2 AHCT. 8% of patients had all APBHC infused during their first AHCT, 83.5% had half of their APBHC infused during their first AHCT, and 8.1% had a different amount of APBHC infused during their first AHCT thus 91.6% of patients who collected APBHC had cells in storage. The median OS for the entire cohort after AHCT was 94.7 months (95% CI 88.6-100). Out of 347 patients who had stem cells in cryopreservation, 5 (1.4%) underwent a salvage AHCT and 3 (0.9%) underwent a tandem AHCT. 61% percent of patients had ≥1 excess collection sessions for APBHC that ultimately went unused. The median cost of excess collection sessions was $4,680 per pt (range, $4,680-$32,760) and the median total cost for excess collection sessions plus costs for storage was $23,840 per pt (range, $4,680-$85,450). The sum of costs of excess collection sessions was $2,077,920 and the sum of costs of cryopreservation was $5,812,665.
Conclusion: The results of this single center analysis show that only 1.4% of patients underwent a salvage AHCT between 2010 and 2019 while 91.6% had APBHC left in storage which confirms the increase in storage of cryopreserved APBHC and declining trend in the use of stored APBHC for salvage AHCT in pts with MM. This is likely due to the advent of next generation novel therapies such as monoclonal antibodies, proteasome inhibitors and immunomodulatory agents. Excess collections, and cryopreservation of unused APBHC incur a cost of nearly $8 million. Institutional policies regarding universal APBHC collection and long-term storage should be reevaluated.
1Phipps C, et al. Bone Marrow Transplantation 2015:50;663-667.
Murthy: CRISPR Therapeutics: Research Funding. Ailawadhi: Sanofi: Consultancy; Medimmune: Research Funding; Genentech: Consultancy; Ascentage: Research Funding; Pharmacyclics: Consultancy, Research Funding; Xencor: Research Funding; Janssen: Consultancy, Research Funding; Amgen: Consultancy, Research Funding; AbbVie: Consultancy; Beigene: Consultancy; GSK: Consultancy, Research Funding; BMS: Consultancy, Research Funding; Takeda: Consultancy; Cellectar: Research Funding; Karyopharm: Consultancy.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal